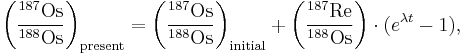Rhenium-osmium dating
Rhenium-Osmium dating is a form of radiometric dating based on the beta decay of the isotope 187Re to 187Os. This normally occurs with a half-life of 41.6 × 109 y,[1] but studies using fully ionised 187Re atoms have found that this can decrease to only 33 y.[2] Both rhenium and osmium are strongly siderophilic (iron loving) and chalcophilic (sulfur loving) making them useful in dating sulfide ores such as gold and Cu-Ni deposits.
This dating method is based on an isochron calculated based on isotopic ratios measured using N-TIMS (Negative – Thermal Ionization Mass Spectrometry).
Rhenium-Osmium isochron
Rhenium-Osmium dating is carried out by the isochron dating method. Isochrons are created by analysing several samples believed to have formed at the same time from a common source. The Re-Os isochron plots the ratio of radiogenic 187Os to non-radiogenic 188Os against the ratio of the parent isotope 187Re to the non-radiogenic isotope 188Os. The stable and relatively abundant osmium isotope 188Os is used to normalize the radiogenic isotope in the isochron.
The Re-Os isochron is defined by the following equation:
where:
- t is the age of the sample,
- λ is the decay constant of 187Re,
- (eλt-1) is the slope of the isochron which defines the age of the system.
A good example of an application of the Re-Os isochron method is a study on the dating of a gold deposit in the Witwatersrand mining camp, South Africa.[3]
References
- ^ Smoliar, M.I.; Walker, R.J.; Morgan, J.W. (1996). "Re-Os ages of group IIA, IIIA, IVA, and IVB iron meteorites". Science 271 (5252): 1099–1102. doi:10.1126/science.271.5252.1099.
- ^ Bosch, F.; Faestermann, T.; Friese, J.; Heine, F.; Kienle, P.; Wefers, E.; Zeitelhack, K.; Beckert, K. et al. (1996). "Observation of bound-state β– decay of fully ionized 187Re:187Re-187Os Cosmochronometry". Physical Review Letters 77 (26): 5190–5193. Bibcode 1996PhRvL..77.5190B. doi:10.1103/PhysRevLett.77.5190. PMID 10062738.
- ^ Kirk, J.; Ruiz, J.; Chesley, J.; Walshe, J.; England, G. (2002). "A major Archean, gold- and crust-forming event in the Kaapvaal Craton, South Africa". Science 297 (5588): 1856–1858. doi:10.1126/science.1075270. PMID 12228713.